As part of a continuing coordinated response to coronavirus (COVID-19), Governor Pritzker has issued a disaster proclamation to allow Illinois to access additional federal resources. The proclamation formalizes emergency procedures already underway across the state by activating the State Emergency Operations Center (SEOC) to deploy other resources to address the issue.
The Illinois Department of Public Health (IDPH) is currently operating three testing labs statewide – in Chicago, Springfield and Carbondale. These state labs meet current need, and commercial testing expansion is anticipated this week.
Major health insurance companies in Illinois, including Blue Cross and Blue Shield of Illinois, UnitedHealthcare, Aetna and Cigna have announced they will waive the costs for COVID-19 testing. Medicaid and Medicare are also covering testing costs.
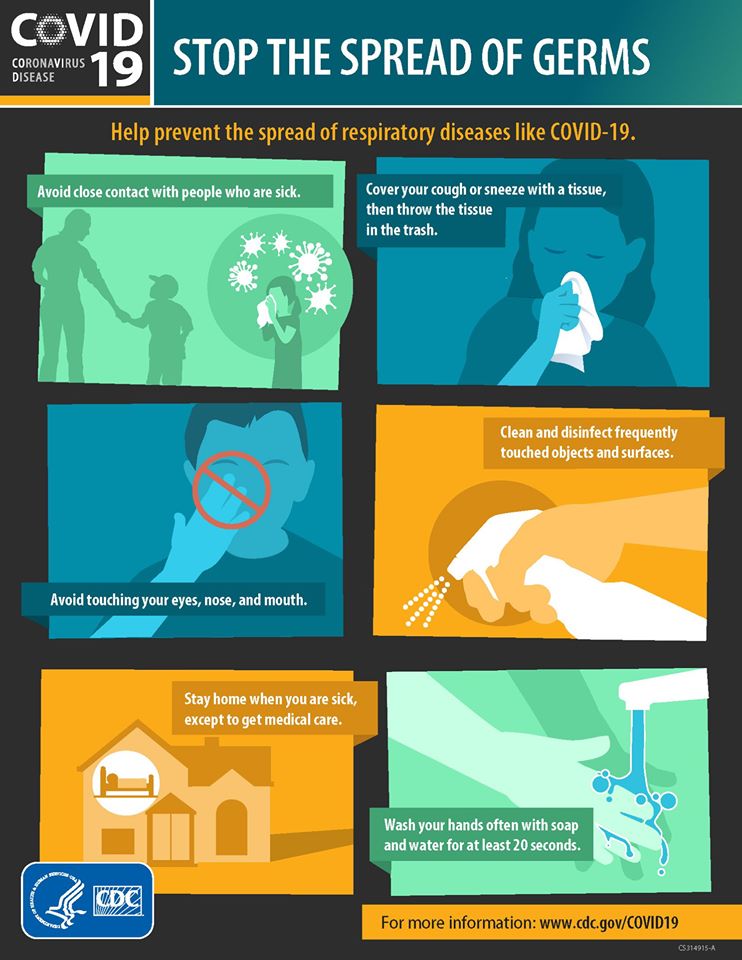
Public health officials encourage the public to follow the same precautions as flu season to help prevent the spread of COVID-19:
- Wash your hands frequently with soap and water for at least 20 seconds;
- Cover your cough or sneeze;
- Avoid touching your eyes, nose and mouth;
- Clean often touched surfaces frequently;
- Maintain social distance (3 feet) between yourself and anyone who is coughing or sneezing;
- Avoid visiting long-term care facilities, such as nursing homes, especially if you’re sick;
- Stay home if you do not feel well;
- Plan a safe visit to the doctor if you experience any symptoms.
Vulnerable populations – including people 60 years and older or those with certain health conditions like heart disease, lung disease, and weakened immune systems – are recommended to avoid large gatherings.
IDPH has a statewide COVID-19 hotline and website to answer any questions from the public or to report a suspected case: call 1-800-889-3931 or visit IDPH.illinois.gov.
For information about how you, your school, your workplace and your community can prepare, please visit Preventing COVID-19 Spread in Communities.
Below is a series of frequently asked questions and recommendations from the Centers for Disease Control (CDC):
Q: What are the clinical features of COVID-19?
A: The clinical spectrum of COVID-19 ranges from mild disease with non-specific signs and symptoms of acute respiratory illness, to severe pneumonia with respiratory failure and septic shock. There have also been reports of asymptomatic infection with COVID-19. See also Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease 2019 (COVID-19).
Q: Who is at risk for COVID-19?
A: Currently, those at greatest risk of infection are persons who have had prolonged, unprotected close contact with a patient with symptomatic, confirmed COVID-19 and those with recent travel to China, especially Hubei Province.
Q: Who is at risk for severe disease from COVID-19?
The available data are currently insufficient to identify risk factors for severe clinical outcomes. From the limited data that are available for COVID-19 infected patients, and for data from related coronaviruses such as SARS-CoV and MERS-CoV, it is possible that older adults, and persons who have underlying chronic medical conditions, such as immunocompromising conditions, may be at risk for more severe outcomes. See also See also Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease 2019 (COVID-19).
Q: When is someone infectious?
A: The onset and duration of viral shedding and period of infectiousness for COVID-19 are not yet known. It is possible that SARS-CoV-2 RNA may be detectable in the upper or lower respiratory tract for weeks after illness onset, similar to infection with MERS-CoV and SARS-CoV. However, detection of viral RNA does not necessarily mean that infectious virus is present. Asymptomatic infection with SARS-CoV-2 has been reported, but it is not yet known what role asymptomatic infection plays in transmission. Similarly, the role of pre-symptomatic transmission (infection detection during the incubation period prior to illness onset) is unknown. Existing literature regarding SARS-CoV-2 and other coronaviruses (e.g. MERS-CoV, SARS-CoV) suggest that the incubation period may range from 2–14 days.
Q: Which body fluids can spread infection?
A: Very limited data are available about detection of SARS-CoV-2 and infectious virus in clinical specimens. SARS-CoV-2 RNA has been detected from upper and lower respiratory tract specimens, and SARS-CoV-2 has been isolated from upper respiratory tract specimens and bronchoalveolar lavage fluid. SARS-CoV-2 RNA has been detected in blood and stool specimens, but whether infectious virus is present in extrapulmonary specimens is currently unknown. The duration of SARS-CoV-2 RNA detection in upper and lower respiratory tract specimens and in extrapulmonary specimens is not yet known but may be several weeks or longer, which has been observed in cases of MERS-CoV or SARS-CoV infection. While viable, infectious SARS-CoV has been isolated from respiratory, blood, urine, and stool specimens, in contrast – viable, infectious MERS-CoV has only been isolated from respiratory tract specimens. It is not yet known whether other non-respiratory body fluids from an infected person including vomit, urine, breast milk, or semen can contain viable, infectious SARS-CoV-2.
Q: Can people who recover from COVID-19 be infected again?
A: The immune response to COVID-19 is not yet understood. Patients with MERS-CoV infection are unlikely to be re-infected shortly after they recover, but it is not yet known whether similar immune protection will be observed for patients with COVID-19.
Q: How should healthcare personnel protect themselves when evaluating a patient who may have COVID-19?
A: Although the transmission dynamics have yet to be determined, CDC currently recommends a cautious approach to persons under investigation (PUI) for COVID-19. Healthcare personnel evaluating PUI or providing care for patients with confirmed COVID-19 should use Standard Precautions, Contact Precautions, Airborne Precautions, and use eye protection (e.g., goggles or a face shield). See the Interim Infection Prevention and Control Recommendations for Patients with Known or Patients Under Investigation for Coronavirus Disease 2019 (COVID-19) in Healthcare Settings.
Q: Should any diagnostic or therapeutic interventions be withheld due to concerns about transmission of COVID-19?
A: Patients should receive any interventions they would normally receive as standard of care. Patients with suspected or confirmed COVID-19 should be asked to wear a surgical mask as soon as they are identified and be evaluated in a private room with the door closed, ideally an airborne infection isolation room, if available. Healthcare personnel entering the room should use Standard Precautions, Contact Precautions, Airborne Precautions, and use eye protection (e.g., goggles or a face shield).
Q: How do you test a patient for SARS-CoV-2, the virus that causes COVID-19?
A: See recommendations for reporting, testing, and specimen collection at Interim Guidance for Healthcare Professionals.
Q: Will existing respiratory virus panels, such as those manufactured by Biofire or Genmark, detect SARS-CoV-2, the virus that causes COVID-19?
A: No. These multi-pathogen molecular assays can detect a number of human respiratory viruses, including other coronaviruses that can cause acute respiratory illness, but they do not detect COVID-19.
Q: How is COVID-19 treated?
Not all patients with COVID-19 will require medical supportive care. Clinical management for hospitalized patients with COVID-19 is focused on supportive care of complications, including advanced organ support for respiratory failure, septic shock, and multi-organ failure. Empiric testing and treatment for other viral or bacterial etiologies may be warranted.
Corticosteroids are not routinely recommended for viral pneumonia or ARDS and should be avoided unless they are indicated for another reason (e.g., COPD exacerbation, refractory septic shock following Surviving Sepsis Campaign Guidelines).
There are currently no antiviral drugs licensed by the U.S. Food and Drug Administration (FDA) to treat COVID-19. Some in-vitro or in-vivo studies suggest potential therapeutic activity of some agents against related coronaviruses, but there are no available data from observational studies or randomized controlled trials in humans to support recommending any investigational therapeutics for patients with confirmed or suspected COVID-19 at this time. Remdesivir, an investigational antiviral drug, was reported to have in-vitro activity against COVID-19. A small number of patients with COVID-19 have received intravenous remdesivir for compassionate use outside of a clinical trial setting. A randomized placebo-controlled clinical trial of remdesivirexternal icon for treatment of hospitalized patients with COVID-19 respiratory disease has been implemented in China. A randomized open label trialexternal icon of combination lopinavir-ritonavir treatment has been also been conducted in patients with COVID-19 in China, but no results are available to date. trials of other potential therapeutics for COVID-19 are being planned. For information on specific clinical trials underway for treatment of patients with COVID-19 infection, see clinicaltrials.govexternal icon.
Q: Should post-exposure prophylaxis be used for people who may have been exposed to COVID-19?
A: There is currently no FDA-approved post-exposure prophylaxis for people who may have been exposed to COVID-19. For more information on movement restrictions, monitoring for symptoms, and evaluation after possible exposure to COVID-19 See Interim US Guidance for Risk Assessment and Public Health Management of Persons with Potential Coronavirus Disease 2019 (COVID-19) Exposure in Travel-associated or Community Settings and Interim U.S Guidance for Risk Assessment and Public Health Management of Healthcare Personnel with Potential Exposure in a Healthcare Setting to Patients with Coronavirus Disease 2019 (COVID-19).
Q: Whom should healthcare providers notify if they suspect a patient has COVID-19?
A: Healthcare providers should consult with local or state health departments to determine whether patients meet criteria for a Persons Under Investigation (PUI). Providers should immediately notify infection control personnel at their facility if they suspect COVID-19 in a patient.
Q: Do patients with confirmed or suspected COVID-19 need to be admitted to the hospital?
A: Not all patients with COVID-19 require hospital admission. Patients whose clinical presentation warrants in-patient clinical management for supportive medical care should be admitted to the hospital under appropriate isolation precautions. Some patients with an initial mild clinical presentation may worsen in the second week of illness. The decision to monitor these patients in the inpatient or outpatient setting should be made on a case-by-case basis. This decision will depend not only on the clinical presentation, but also on the patient’s ability to engage in monitoring, the ability for safe isolation at home, and the risk of transmission in the patient’s home environment. For more information, see Interim Infection Prevention and Control Recommendations for Patients with Known or Patients Under Investigation for Coronavirus Disease 2019 (COVID-19) in a Healthcare Setting and Interim Guidance for Implementing Home Care of People Not Requiring Hospitalization for Coronavirus Disease 2019 (COVID-19).
Q: When can patients with confirmed COVID-19 be discharged from the hospital?
A: Patients can be discharged from the healthcare facility whenever clinically indicated. Isolation should be maintained at home if the patient returns home before the time period recommended for discontinuation of hospital Transmission-Based Precautions described below.
Decisions to discontinue Transmission-Based Precautions or in-home isolation can be made on a case-by-case basis in consultation with clinicians, infection prevention and control specialists, and public health based upon multiple factors, including disease severity, illness signs and symptoms, and results of laboratory testing for COVID-19 in respiratory specimens.
Criteria to discontinue Transmission-Based Precautions can be found in: Interim Considerations for Disposition of Hospitalized Patients with COVID-19
Q: What do I need to know if a patient with confirmed or suspected COVID-19 asks about having a pet or other animal in the home?
A: See COVID-19 and Animals.